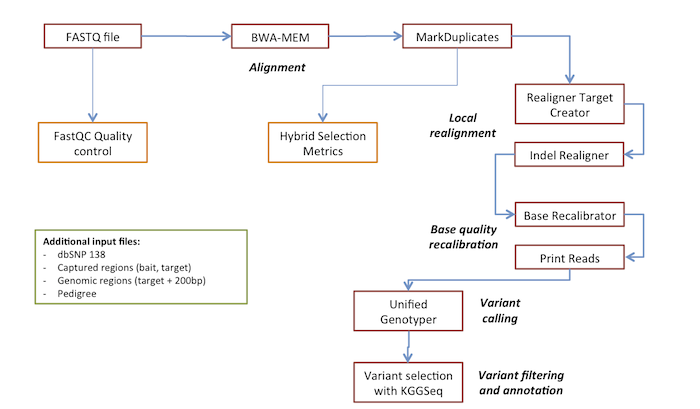
Library prep on 50ng of genomic DNA was performed using the Twist Human Core Exome kit (Twist Bioscience, South San Francisco, CA, USA) according to the manufacturer’s instructions on a Hamilton STAR robot (Hamilton, Bonaduz, Switzerland). Genomic DNA was extracted from either amniotic fluid, chorion villi or cultured amniocytes using the Maxwell RSC Blood DNA kit on a Maxwell RSC 48 Instrument (Promega, Madison, MI, USA). 
Here we discuss the opportunities and challenges for the use of WES in the diagnosis of fetuses with ultrasound abnormalities and provide suggestions for implementation of this valuable technique in other labs. Recently, a national framework has been formulated guiding the indication, analysis and reporting of prenatal WES. For all indications, a genome-wide microarray is performed with national consensus guidelines in place steering the interpretation and reporting of the results. In Belgium, the molecular analysis of publicly funded invasive prenatal diagnosis can only be executed at one of the eight Centers for Medical Genetics. This paper describes the experiences of a single Belgian genetic center with the implementation of WES in the prenatal diagnostic workflow. With increasing evidence of the relevance of WES in the prenatal context, revision of the guidelines of the International Society for Prenatal Diagnosis (ISPD) offers directions on how to implement it. Several recent metaanalyses have demonstrated an added diagnostic yield of 1.8–68% for prenatal whole exome sequencing (WES), with the yield largely depending on the inclusion criteria and organ system affected. In up to 40% of pregnancies with a fetal structural anomaly, CMA is able to diagnose an aneuploidy or CNV, still leaving more than half of the cases undiagnosed. The etiology is heterogeneous, ranging from prenatal infections over teratologic agents to genetic causes.Ĭhromosomal microarray analysis (CMA) has been widely implemented in the analysis of invasively obtained prenatal samples (amniotic fluid or chorion villi) for the genome-wide detection of both aneuploidies and microdeletions/microduplications (copy number variants or CNVs). Major congenital anomalies (MCA) have a prevalence of 2–3% and are responsible for a significant percentage of pre- and perinatal demise and neonatal morbidity. With a diagnostic yield in selected cases of 25% and a turn-around time under 4 weeks, rapid WES shows promise for becoming part of pregnancy care in fetuses with ultrasound anomalies in whom chromosomal microarray did not uncover the cause. Prenatal rapid WES allows for a timely decision-making in the current pregnancy, adequate counseling with the possibility of preimplantation or prenatal genetic testing in future pregnancies and screening of the extended family. Autosomal recessive (4), de novo (2) and dominantly inherited (1) mutations were detected. Twenty-eight fetus-parent trios were analyzed, of which seven (25%) showed a pathogenic or likely pathogenic variant that explained the fetal phenotype. We present the results of 1 year of prenatal WES in a single genetic center. In the last years, WES is slowly being implemented in the prenatal setting as well, although some hurdles remain, such as quantity and quality of input material, minimizing turn-around times, and ensuring consistent interpretation and reporting of variants. Whole exome sequencing (WES) has become part of the postnatal diagnostic work-up of both pediatric and adult patients with a range of disorders.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
